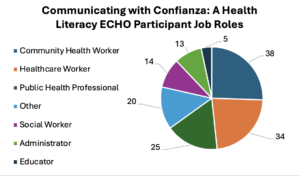
San Antonio, Texas — An individual’s health literacy — the ability to find, understand and use health information — can vary during one’s life. Most all of us will find ourselves needing support at one point or another.
Because of that, Health Confianza’s co-director Jason Rosenfeld wanted to train community members to serve as Ambassadors, providing health information and local resources to community members where and when they need it.
Enter the Confianza Ambassador Program with the goal of creating a bridge between healthcare and the community. To begin, Health Confianza and its partners identified 15 trusted and respected community leaders who were motivated to serve as community educators and resources for the greater community.
The Confianza Ambassadors were selected for their ability to be local champions, people who can build connections with hard-to-reach populations and create innovative health messaging.
Over a period of months, the Ambassadors were trained to deliver scientifically accurate preventive health information, that is both culturally and linguistically appropriate. The Ambassadors would meet weekly to receive training on topics including teach-back (a method for sharing health information) and community outreach.
There were eight in-person sessions followed by monthly check-in meetings to discuss upcoming opportunities, challenges and share resources.
Stand-out Ambassador
One Ambassador, Jessica Route, saw the program as a great match for her life experiences and personal desire to serve.
Route was approached with the opportunity to become a Health Confianza Ambassador while she was working for WestCare Texas in its Americorp program as an HIV peer navigator. Under WestCare Texas, she also received her medical assistant and phlebotomy certifications last year. This year, she moved into a position as an Opiate Impacted Family Support Navigator.

“I like being impactful and helping community members. My life’s ambition is to give back to the community, so I’ve seen it as an opportunity to not only learn for myself but also to help others,” Route said.
Route’s experience as an HIV navigator motivated her to want to improve her skills in communicating complex health issues and resources.
“I suppose it’s the lack of education that causes a stigma around HIV, because of what they don’t know. But if they understood better, there would be less for them to fear,” she said. “Not everyone is on the same level. There are barriers and sometimes it just takes being able to explain to them the simplest things, so they’ll understand the bigger things.”
Route said going through the Ambassador program, including delivering a final presentation, helped to boost her confidence in working with people who are living with HIV.
Along with her work with WestCare, she considers herself an “alumna” of Corazon Ministries, which helps houseless individuals in addiction with services, food, housing and other resources. Route stays active with Corazon Ministries and volunteers at Travis Park Church.
In her work as an Ambassador, Route engaged in a mix of activities, from creating social media posts and flyers to attending events and having one-on-one conversations with community. She enjoyed creating a flyer on respiratory illnesses from scratch, which included an explanation of how the respiratory system works.
“What Health Confianza did was educate and inform me, and I was able to share some of that with the community,” she said.
As part of the program, Ambassadors met regularly to share information and work through challenges.
“When I go to a meeting, I feel like we can narrow down (an issue) and get on the same page. I also appreciate sharing resources. There are some many resources that people don’t know about it. They may not even use the internet for whatever reason, so word of mouth is how they learn about things,” she said.
The Ambassador program is “the opportunity to be a bridge. To connect people to resources and information services that they may not know is available. And it feels powerful to be that link between confusion and clarity,” Route said.


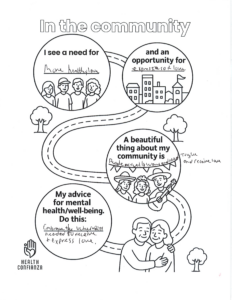
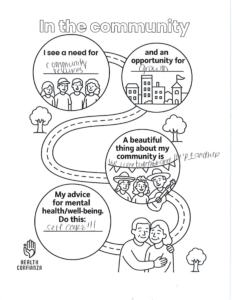
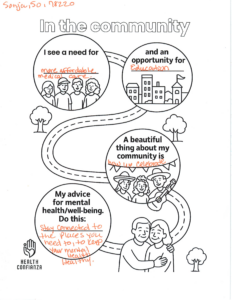
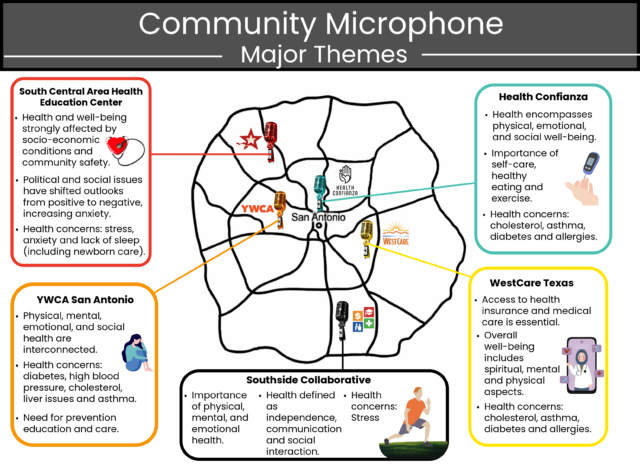
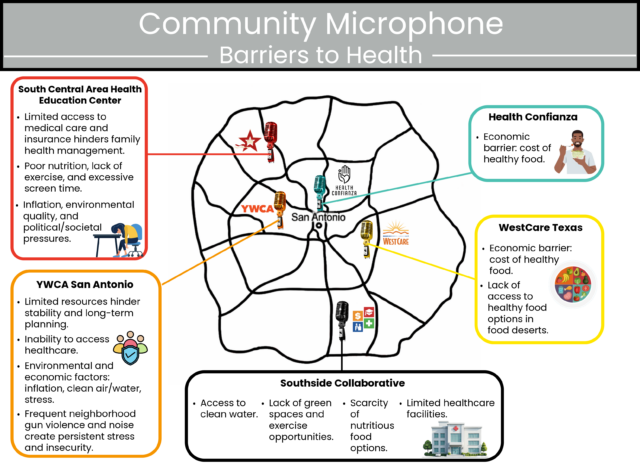
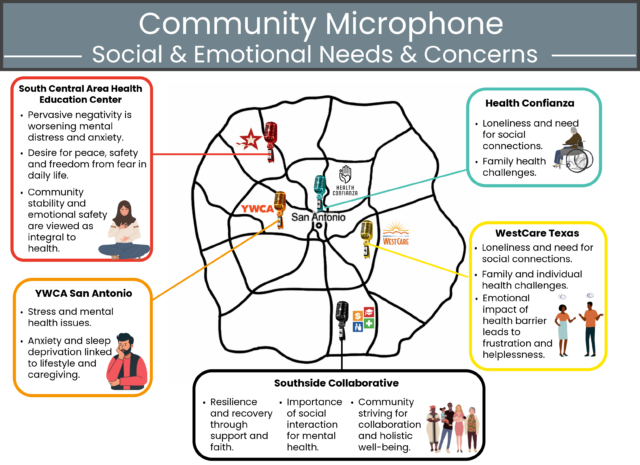
 San Antonio, TEXAS — Health Confianza and its main partners – South Central Area Health Education Center (AHEC), Southside Collaborative, YWCA San Antonio and WestCare Texas – Ella Austin – celebrated the end of a two-year grant at the Building Confianza: Voices, Lesson and Actions health literacy summit on May 15th at Texas A&M San Antonio. .
San Antonio, TEXAS — Health Confianza and its main partners – South Central Area Health Education Center (AHEC), Southside Collaborative, YWCA San Antonio and WestCare Texas – Ella Austin – celebrated the end of a two-year grant at the Building Confianza: Voices, Lesson and Actions health literacy summit on May 15th at Texas A&M San Antonio. .